Welcome to WordPress. This is your first post. Edit or delete it, then start writing!
Why America's Top Doctors Just Broke Away From the CDC?

In a significant development that could reshape how Americans receive and perceive vaccine information, the American Medical Association (AMA) has announced the establishment of an independent vaccine safety and effectiveness review system. This groundbreaking initiative represents a notable shift in how vaccine science is communicated to both healthcare providers and the public.
A Response to Growing Concerns
The launch of this independent review system comes at a critical juncture. In recent years, the Centers for Disease Control and Prevention (CDC) has faced mounting criticism regarding its vaccine evaluation processes, with concerns ranging from transparency issues to questions about potential conflicts of interest. The AMA’s decision to create a parallel review mechanism acknowledges these concerns while offering a fresh approach to vaccine science assessment.
“The timing is particularly significant as public confidence in traditional health institutions has experienced notable fluctuations, especially following the COVID-19 pandemic. By establishing an independent review framework, the AMA appears to be addressing the need for multiple, credible sources of vaccine information that physicians and patients can rely upon.”


Starting With the Essentials: Flu, COVID-19, and RSV
The AMA has chosen to focus its initial efforts on three vaccines that represent some of the most widely administered and discussed immunizations in current medical practice:
Influenza Vaccines: Annual flu shots remain one of the most common preventive health measures, yet vaccination rates have struggled to reach optimal levels. An independent review of flu vaccine effectiveness could provide healthcare providers with additional data to inform their recommendations.
COVID-19 Vaccines: Perhaps no medical intervention in modern history has generated more public debate than COVID-19 vaccines. An independent assessment of their safety profile and effectiveness across different populations and timeframes could contribute valuable perspectives to ongoing discussions.
RSV Vaccines: As newer additions to the vaccine landscape, particularly for older adults and pregnant women, RSV vaccines represent an area where independent review could help establish clearer understanding of benefits and considerations.
What Independence Really Means
The term “independent” is crucial here. While the AMA has not released complete details about the review system’s methodology, the emphasis on independence suggests several key features:
- Separate from federal health agencies: The review process will operate distinctly from CDC evaluations, potentially offering different analytical perspectives
- Physician-focused: As an organization representing doctors, the AMA’s reviews are likely to emphasize clinical relevance and practical application
- Transparent methodology: Independence implies clear disclosure of review processes, funding sources, and potential conflicts of interest
Implications for Healthcare Providers
For physicians and other healthcare providers, this development offers several potential advantages:
Enhanced Decision-Making Tools: Having multiple sources of rigorously reviewed vaccine data allows clinicians to make more informed recommendations tailored to individual patient needs.
Addressing Patient Questions: When patients express concerns about vaccines or ask for “second opinions” on vaccine science, providers will have an additional authoritative resource to reference.
Professional Autonomy: Independent reviews may help physicians feel more confident in their clinical judgment, particularly when navigating complex or contentious vaccine discussions with patients.
The Broader Context: Trust in Medicine
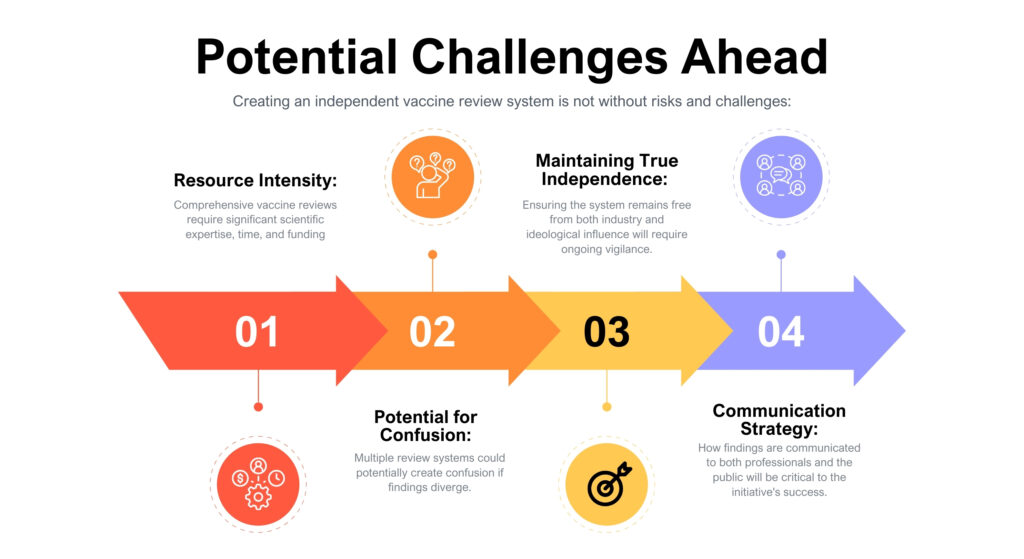
This initiative reflects broader conversations about trust, transparency, and institutional authority in American healthcare. The AMA’s move suggests an understanding that in an era of information abundance and institutional skepticism, multiple credible voices may serve public health better than singular authority.
However, the success of this review system will depend heavily on execution. Questions remain about funding, the composition of review panels, conflict-of-interest policies, and how findings will be communicated.
What This Means for Patients
For patients and the general public, the AMA’s independent review system could provide:
- Additional Perspective: Another trusted source of information when making vaccine decisions
- Increased Transparency: Potentially more accessible explanations of vaccine science
- Physician Confidence: Doctors equipped with multiple sources of high-quality data may provide more nuanced, personalized guidance


Looking Forward
The establishment of this independent review system represents an acknowledgment that the landscape of medical information and public trust has evolved. Whether this initiative strengthens vaccine confidence or introduces new complexities will depend on the rigor of the reviews, the transparency of the process, and the quality of communication surrounding the findings.
As the AMA begins its work on flu, COVID-19, and RSV vaccines, the medical community and public health advocates will be watching carefully. If successful, this model could expand to other vaccines and potentially influence how medical science is reviewed and communicated more broadly.
In an era where trust in institutions must be earned rather than assumed, the AMA’s independent vaccine review system represents an important experiment in medical transparency and professional accountability. The ultimate measure of success will be whether it helps physicians provide better care and helps patients make more informed decisions about their health.
